Localization Of Medical Equipment: Little Giant WeMed Medical Leverages High-end Fields
With the proposal of the "Healthy China 2030" Planning Outline, the development of China's medical device industry has entered a golden period. This is due to the assistance and blessing of the large domestic market and the global supply system, and is also closely related to the hard work of local companies. Under the equipment upgrade and industrial integration, some tracks in the high-end equipment field have undergone substantial changes, and representative enterprises have also emerged.

Looking at the domestic market, it seems that it has become the creed of local medical device companies to continuously break through technological boundaries. The development of the industry is traceable, and the breakthrough of high-end equipment cannot be achieved overnight. It tests how companies can form an ecological closed loop with their own technologies, how to implement new products vertically, and how to be widely recognized by the market.
Behind the high-end domestic medical equipment, there is now greater significance and thrust. The "14th Five-Year" Medical Equipment Industry Development Plan has become the preliminary formation of comprehensive support capacity for public health and medical health needs.

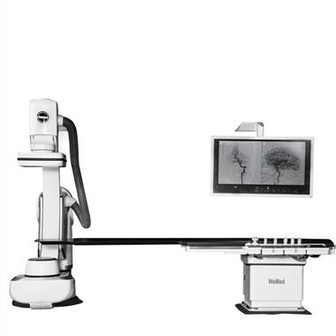
Among them, in the field of interventional diagnosis and treatment equipment, WeMed Medical has built a product matrix with DSA as the core, actively deployed the entire interventional market, and achieved major breakthroughs in DSA and surgical robots, which has brought a subversive impact to the industry.
At the upcoming CMEF 2022, WeMed Medical is going to launch new DSA and solutions. As we can tell, in 2023, WeMed Medical will launch a variety of flat panels of different sizes, suitable for more types of operations such as cardiac and peripheral, and launch dual-tablet DSA and suspended DSA for hybrid operating room models. Combined with an interventional surgical robot and AI image processing platform, a medical solution integrating "Eye, Hand, and Brain" is presented and applied in clinical practice.
With the development of medical technology, the precision of surgery is also increasing. Among them, interventional therapy has the advantages of being precise and minimally invasive and is gradually being valued by many doctors and patients. However, interventional therapy also has its drawbacks. Doctors need to be exposed to X-rays for a long time. Although doctors can perform surgery by wearing lead clothes, radiation can still seriously affect the health of doctors.

Adhering to the concept of "Innovation for Health" for creating brand new domestic products. It is worth mentioning that in the field of vascular interventional surgery robot, WeMed Medical has achieved many technological breakthroughs. The ETcath vascular intervention robot, is completely domestically developed and self-developed. It has obtained more than 100 international and domestic patents.
In 2021, the first PCI operation was completed with ETcath by Professor Zhou Yujie’s team in Beijing Anzhen Hospital. The ETcath is the first domestic self-developed vascular interventional surgery robot that supports multiple types of catheters. It was the first clinical case of balloon dilatation and coronary stent implantation using the domestic vascular interventional robot---ETcath.
As Professor Zhou said, assisted by ETcath, It can not only make the surgery more precise and reduce the complications of patients, but also avoid the damage of X-ray radiation. During the whole process, guide wire manipulation, balloon delivery, stent delivery, and IVUS catheter push were all completed by ETcath.
All these achievements are inseparable from WeMed Medical's strict control of product quality and control of its own product quality with the highest international standards. Hire industry experts as CMO, continue to develop scientific research capabilities, obtain clinical advice more efficiently, and more clearly define the "clinical-centered" development direction in medical equipment research and development, clinical application, etc.